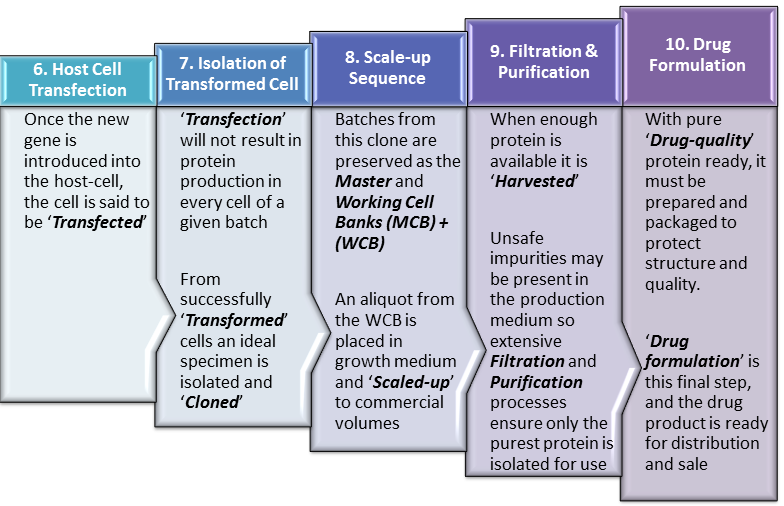
Continuing from our last post, ‘Biopharmaceutical Manufacturing 1‘, this current post, ‘Biopharmaceutical Manufacturing 2‘, elaborates upon the steps in biopharmaceutical manufacture after insertion of the ‘gene of interest‘ into the ‘Expression Vector‘. We therefore begin this post at Step 6 of our Manufacturing Process Flow in which the ‘Vector‘ transports our gene of interest into the host cell. This is presented in even greater detail … Read More
Biopharmaceutical Manufacturing – Part 1
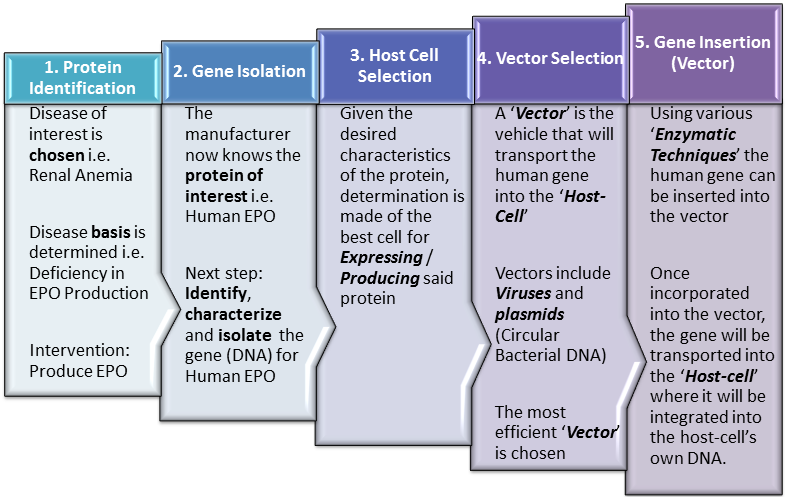
Introduction As noted in earlier posts, because biopharmaceutical manufacturing is dependent upon production within living cells, it is more complex and prone to variability than production of conventional ‘small-molecule‘ / ‘chemical‘ pharmaceuticals. The number of steps involved as well as the complexity of each step, present many opportunities for variability to enter the production process. In fact, minimizing this variability is perhaps the greatest … Read More
What is a Biopharmaceutical? (Part I): The Role of Genetic Engineering
‘ Biopharmaceuticals‘ or Biologics‘ as they are also known are medicines derived from biological sources as suggested by their ‘bio-‘ prefix. Technically this would include, medicines derived by extraction of natural products from plants, animals or even fungi. Penicillins would be one such noted example. Based on this broad definition, vaccines and blood products among others would also be considered … Read More