Introduction pH is a measure of Hydrogen ion concentration and can also be described as ‘A measure of how acidic a solution is‘. Low pH (0-3) environments are most acidic while high pH (11-14) environments are least acidic and described as alkaline or basic. As mentioned previously, protein structure is dependent on a combination of chemical bonds and electrostatic (Stationary … Read More
Biopharmaceutical Storage & Handling: Solutes & Solvents
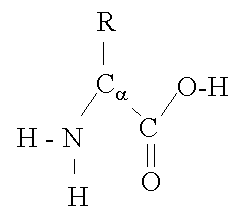
As noted in earlier posts such as ‘Biopharmaceutical Protein Structure – Part 1‘, the elaborate 3-dimensional structure of proteins is maintained by a complex interplay of chemical bonds and electrostatic interactions. These electrostatic interactions primarily involve the charged side-groups of the protein’s constituent amino acids. As seen in Figure 1 below, the ‘R‘ groups which represent clusters of atoms unique to each … Read More
Biopharmaceutical Protein Structure – Part 2
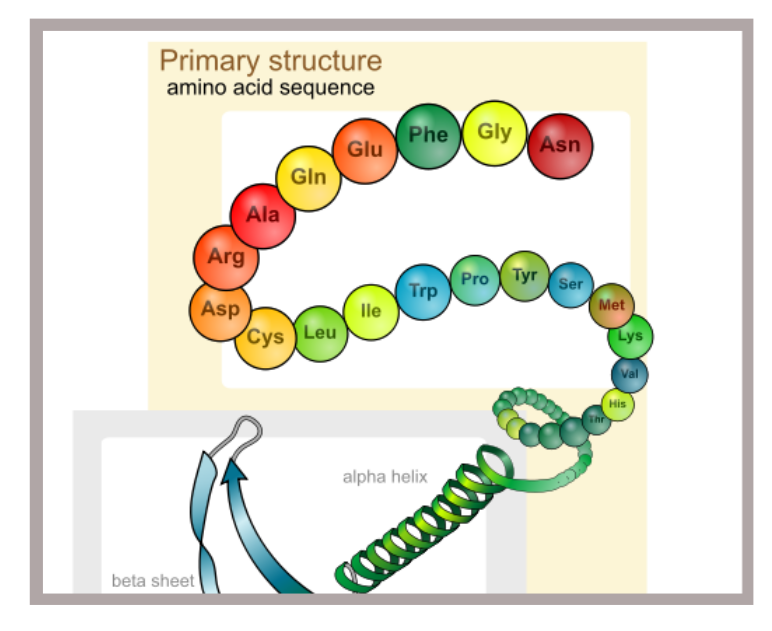
Introduction Our last post entitled, ‘Biopharmaceutical Protein Structure – Part 1‘, looked briefly at the interplay of different chemical bonds common to proteins and their respective contributions to the maintenance of protein structure. We will now apply those principles to the current discussion and explore in greater detail the four levels of protein structure with particular emphasis on the structural complexity of proteins. The Structural Complexity of … Read More
Biopharmaceutical Protein Structure – Part 1
Introduction Given that the overwhelming majority of biopharmaceuticals are proteins, this article will explore some key points on protein structure. This is particularly important because protein function is directly related to protein structure. Specifically we will look at the different types of molecular bonds found in proteins and their relationship to the final three-dimensional configuration of said proteins. If we are to appreciate … Read More